| Home |
| Research |
| Python & Hardware |
| Teaching |
| Publications |
|
|
My research aims to uncover how specific neural circuits support
1. motor control and
2. spatial orientation
in the intact brain.
1. Cerebellum and complex motor control
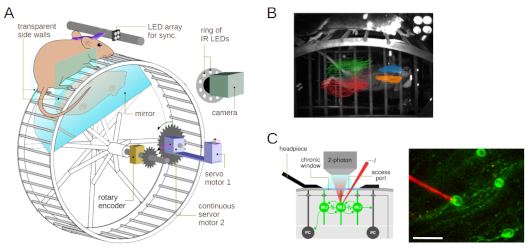
|
| Fig 1. A) To investigated the cerebellar role in skilled limb movements and locomotor learning in mice, we developped a novel task: LocoReach. B) The task allows for highly refined behavioral analysis of locomotor dynamics and precision. C) The task implementation furthermore opens the possibility for neural recordings during task execution. |
Active collaborations in this project:
- N Alex Cayco Gajic (Group for Neural Theory, ENS, Paris): studies network dynamics using simulations, expert in machine learning approaches for large-scale data analysis.
- Heike Stein (ISIR, Sorbonne Université, Paris): studies how natural behavior is generated from neural activity, expert in data-driven modeling of behavioral dynamics as well as large-scale neural data analysis.
2. Multisensory integration in the head-direction system
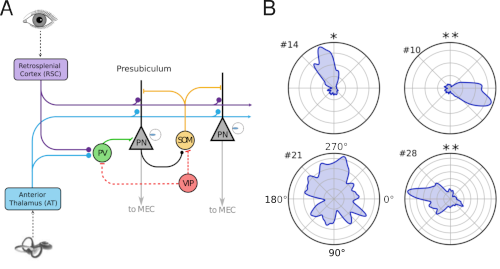
|
| Fig 2. A) We study how vestibular- and visual sensory information is combined by head-direction circuits. We are particularly interested in cell-type-specific roles in input integration. B) Head-direction tuning curves quantifying the directionality of neural activity for four example neurons (* indicate significant directional tuning). Curves were aquired with miniature microscopes in freely behaving mice |
A second research direction investigates how the brain builds a sense of orientation in space—our internal “compass.” We study the head-direction system, a network of brain regions that encodes the direction an animal is facing, focusing on how visual, vestibular (balance), and self-motion signals are integrated in the presubiculum. Using miniature microscopes to image neural activity in freely behaving mice, combined with controlled sensory environments and computational modeling, we examine how populations of neurons maintain stable direction signals and update them when the environment changes. This work aims to reveal general principles of how the brain combines multiple sensory inputs to generate coherent internal representations of the world.
Active collaborations in this project:
- Desdemona Fricker (SPPIN, Université Paris Cité, Paris, France): Desdemona's work investigates how neural circuits encode spatial orientation and head‑direction signals in the brain.
- Lucia Wittner (Research Center for Natural Sciences, Budapest, Hungary): Lucia's research explores how excitatory and inhibitory neuronal networks generate and propagate synchronous activity in the human cortex and hippocampus, with implications for understanding epilepsy.